
A mutation in the prion protein gene resulting in an exchange of aspartate for asparagine at codon 187 (D187N) is responsible for a genetic disease here, but whether the disease phenotype will be FFI (methionine) or genetic CJD (valine) depends on the polymorphism at codon 129 of the affected allele. In patients with a short disease course who died during an earlier stage of the disease, the presence of prion aggregates may be limited to the thalamic and midbrain regions, the entorhinal cortex, and the amygdala. The distribution of pathologic prion protein aggregates is more widespread than the histological lesions characteristic of prion disease, i.e., spongiform changes of the neuropil, neuronal loss, and gliosis. Clinically, the disease course is characterized by progressive untreatable insomnia, dysautonomia, endocrine disbalance, and motor signs, while the pathology is dominated by thalamic degeneration. described FFI as a genetic disease in an Italian family for the first time in 1986. 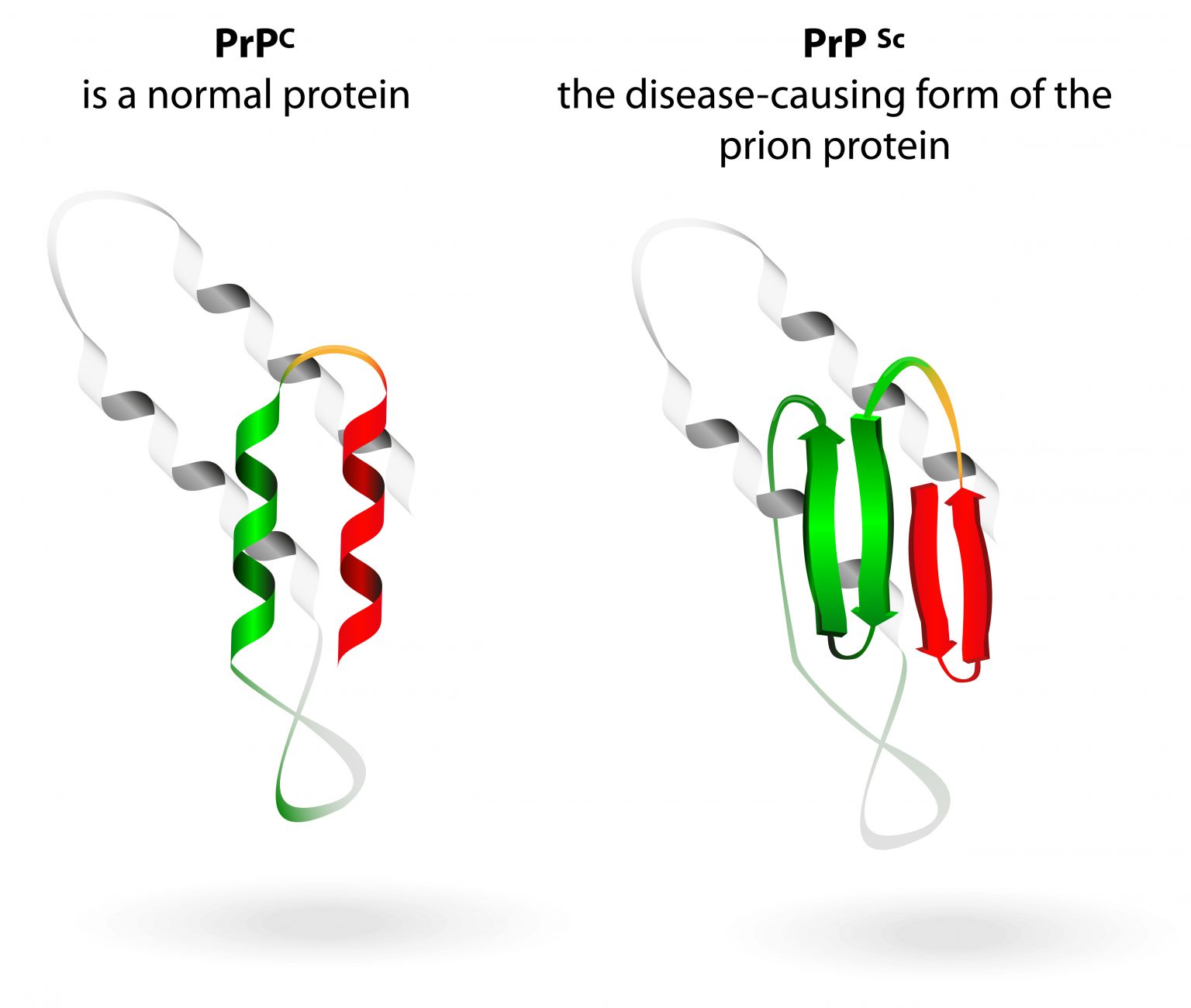
Sporadic, genetic, and acquired forms of prion diseases are known for humans and animals alike. The less soluble pathological isoform tends to form protease-resistant aggregates. The always-fatal TSEs or prion diseases are characterized by their experimental transmissibility, spongiform changes in the central nervous system (CNS), and the infectious agent being a misfolded protein (a proteinaceous infectious particle, abbreviated prion ) that is able to corrupt its physiological counterpart. FFI may resemble a prion type with its own conformation sharing properties partly with type 1 and type 2 prions.įatal familial insomnia (FFI) is a human transmissible spongiform encephalopathy (TSE), such as Creutzfeldt-Jakob disease (CJD) or Gerstmann-Straeussler-Scheinker syndrome (GSS). Additional criteria supplying information about conformational differences or similarities need to be taken into account. Our results suggest that the size of the proteinase-K-resistant fragments is not a valid criterion on its own. Regarding the conformational stability against denaturation with GdnHCl, FFI prion aggregates resembled CJD type 1 more than type 2. While in all cases the unglycosylated fragment in Western blot analysis shared the same size with sporadic CJD prion type 2, the reticular/synaptic deposition pattern of the prion aggregates resembled the ones found in sporadic CJD type 1 (CJD types according to the Parchi classification from 1999). Here, we analyzed the properties of the pathological prion protein in six FFI cases by Western blot analysis, a protein aggregate stability assay, and aggregate deposition characteristics visualized with the paraffin-embedded tissue blot. Fatal familial insomnia (FFI) belongs to the genetic human transmissible spongiform encephalopathies (TSE), such as genetic Creutzfeldt-Jakob disease (CJD) or Gerstmann-Straeussler-Scheinker syndrome (GSS).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
